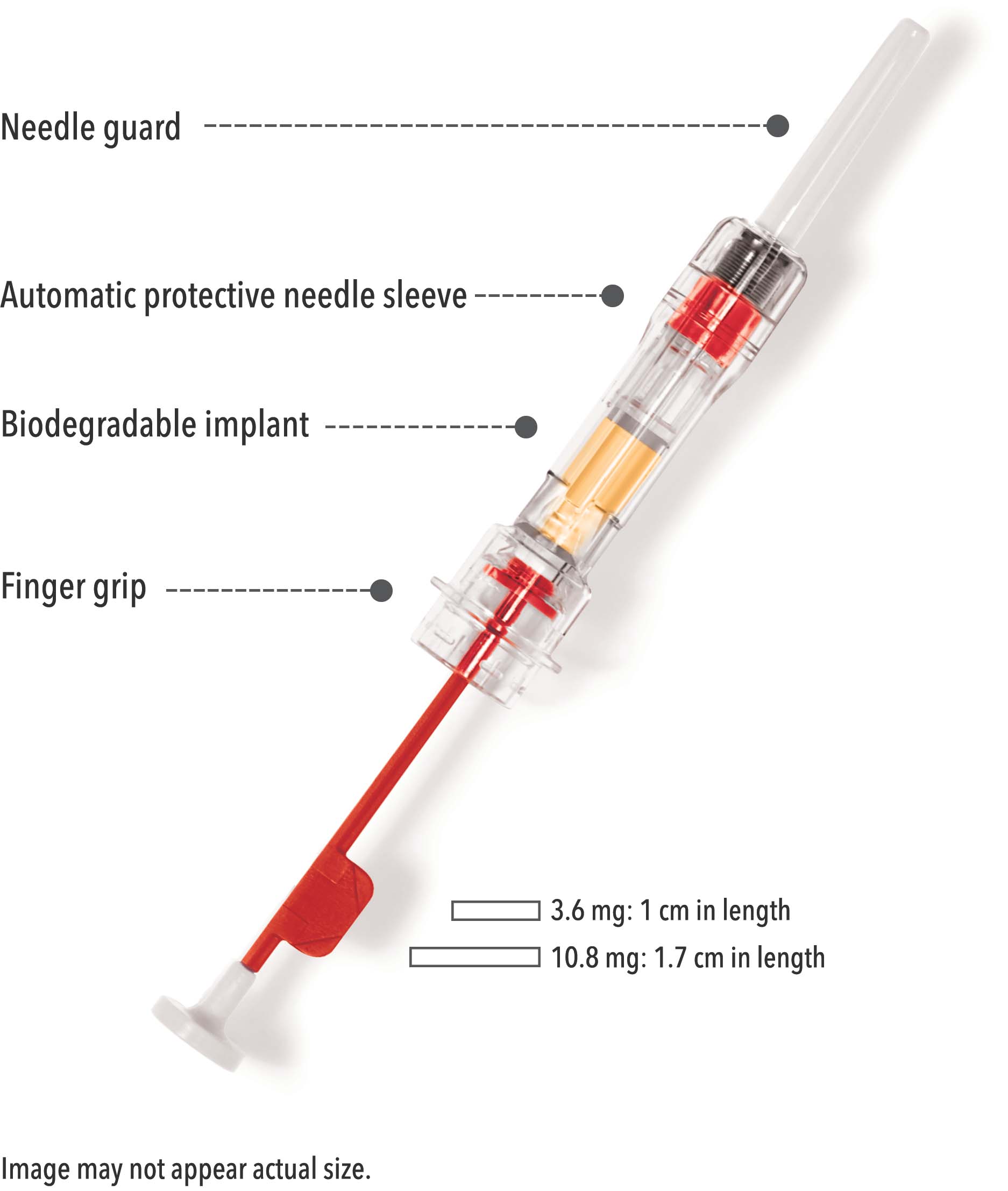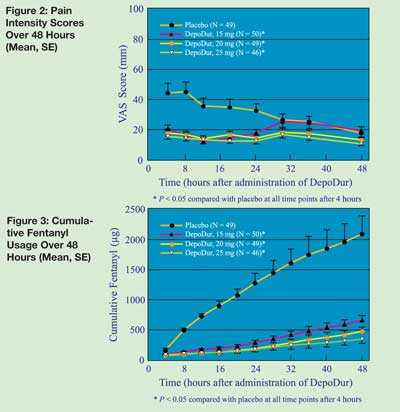
DepoDur™: A New Drug Formulation With Unique Safety Considerations - Anesthesia Patient Safety Foundation

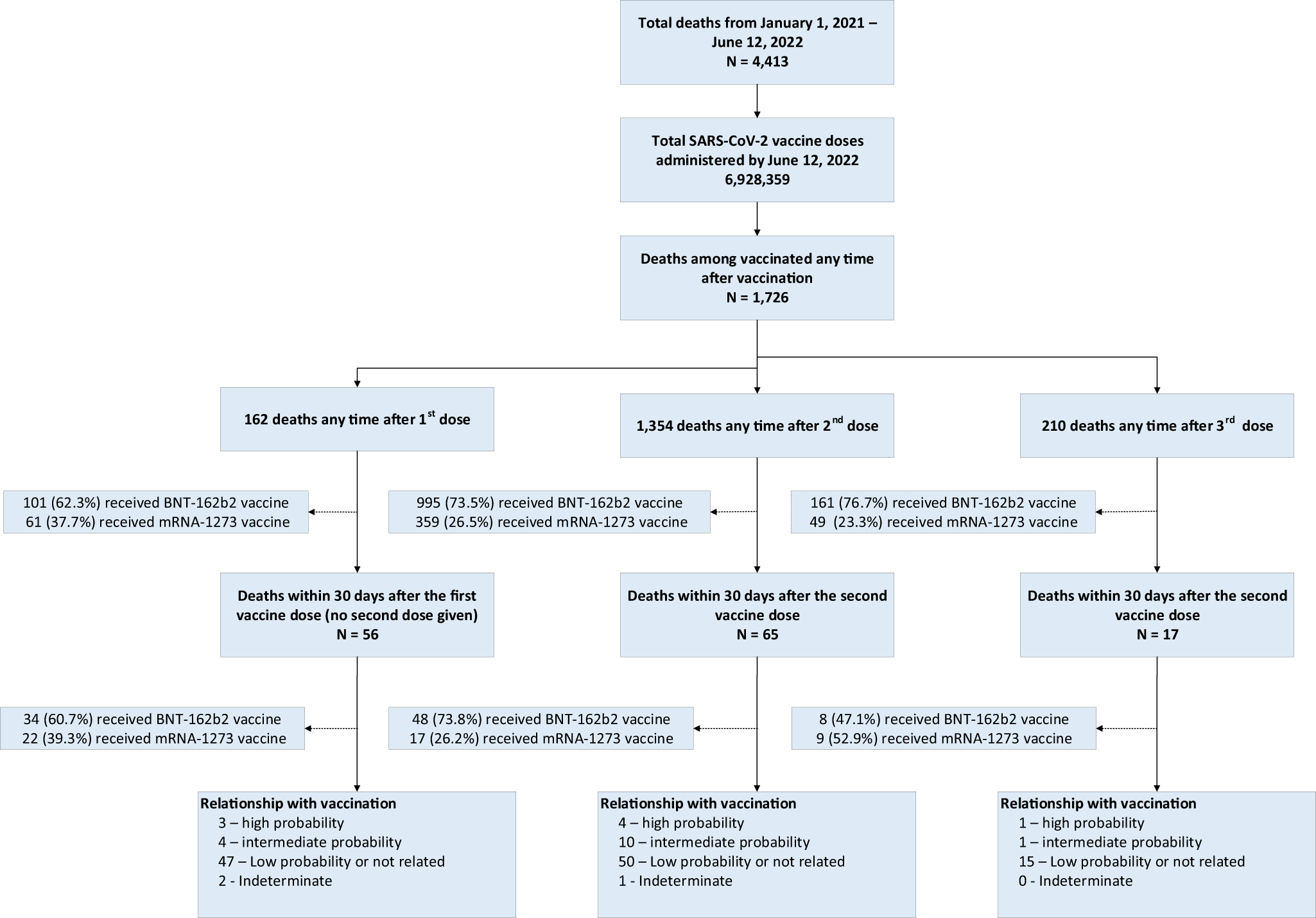
Interim Estimates of Vaccine Effectiveness of BNT162b2 and mRNA-1273 COVID-19 Vaccines in Preventing SARS-CoV-2 Infection Among Health Care Personnel, First Responders, and Other Essential and Frontline Workers — Eight U.S. Locations, December

Lower-dose prescribing: Minimizing “side effects” of pharmaceuticals on society and the environment - ScienceDirect

Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial - The Lancet

Efficacy of a low-dose candidate malaria vaccine, R21 in adjuvant Matrix-M, with seasonal administration to children in Burkina Faso: a randomised controlled trial - The Lancet
Real-World Midazolam Use and Outcomes With Out-of-Hospital Treatment of Status Epilepticus in the United States

Near-Equivalence: Generating Evidence to Support Alternative Cost-Effective Treatments | Journal of Clinical Oncology

Intradermal administration of fractional doses of the inactivated poliovirus vaccine in a campaign: a pragmatic, open-label, non-inferiority trial in The Gambia - The Lancet Global Health
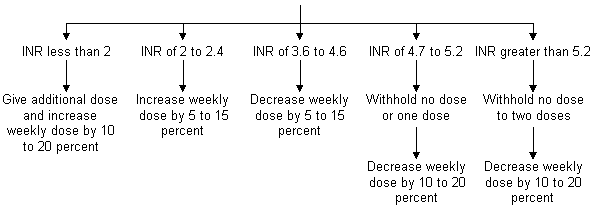
Periprocedural Guidelines for the Management of Patients on long-term Anticoagulant and Antiplatelet Agents
Hepatitis A Vaccination by Intracutaneous Low Dose Administration: A Less Expensive Alternative: Scandinavian Journal of Infectious Diseases: Vol 28, No 5

Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

Parenteral Route: What Is It, Methods of Administration, Advantages, Disadvantages, and More | Osmosis
Assessing the Evidence: Medication Assisted Treatment for Opioid Addiction Dosage and Treatment Limits MAT and Pregnancy Risks